PFS by the Numbers
Adverse drug reaction reports worldwide
PFS research studies published
Known suicides worldwide
Doctors & researchers speaking out
Patient Services
Newsroom
- Brother of UK PFS Patient Who Took His Own Life Speaks out in the Wake of New Finasteride Warning‘We need to continually remind doctors that this drug could prove fatal to any patient,’ says Philip Dixon June 10, 2024 Dear Friends: The British… Read more: Brother of UK PFS Patient Who Took His Own Life Speaks out in the Wake of New Finasteride Warning
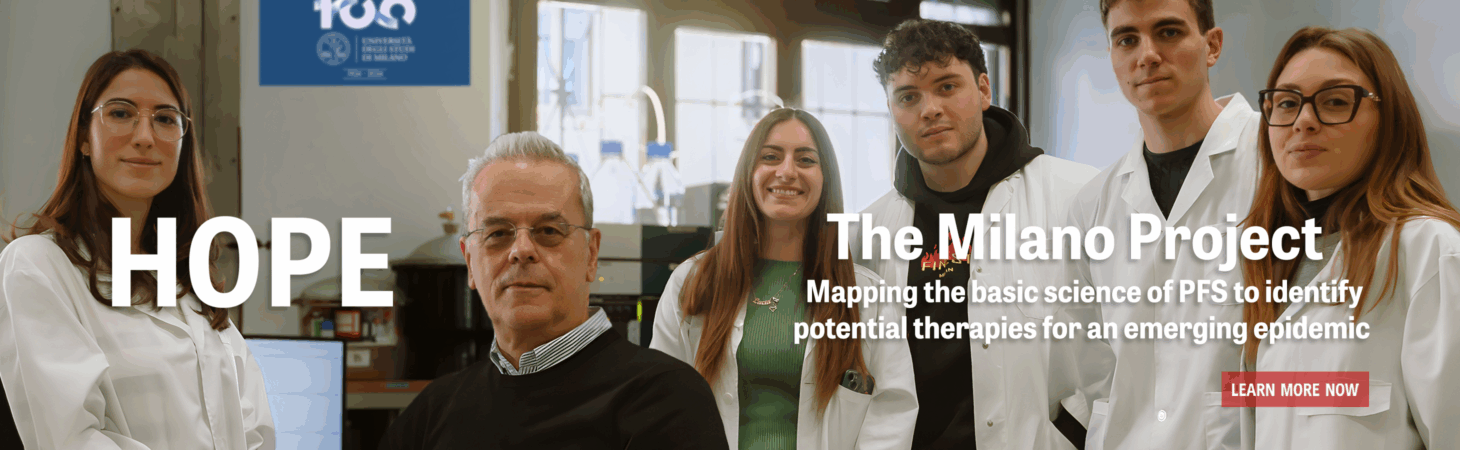
- Team Melcangi Launches ‘Milano Project’ to Map the Basic Science of PFS so Research Can Move from an Animal Model to Human Clinical TrialsInitial therapeutic target is allopregnanolone April 11, 2024 Dear Friends: Roberto Cosimo Melcangi, PhD, is taking a page out of Robert Oppenheimer’s playbook. The Head… Read more: Team Melcangi Launches ‘Milano Project’ to Map the Basic Science of PFS so Research Can Move from an Animal Model to Human Clinical Trials
- Search for Sources of Major PFS Symptoms Pinpoints 186 Brain GenesNew UniMi study enumerates pathologies including depression, anxiety, insomnia, and cognitive dysfunction potentially linked to finasteride-induced genetic dysregulation March 16, 2024 Dear Friends: Turns out,… Read more: Search for Sources of Major PFS Symptoms Pinpoints 186 Brain Genes
- Canada to Health Pros: All Finasteride Patients Should ‘Be Screened for Suicidal Ideation, Self-harm, and Depression’ Before Being Prescribed the DrugJapanese disproportionality analysis of finasteride ADRs, meanwhile, shows completed suicides 270% higher than expected Jan. 28, 2024 Dear Friends: Canada is taking no chances vis-á-vis… Read more: Canada to Health Pros: All Finasteride Patients Should ‘Be Screened for Suicidal Ideation, Self-harm, and Depression’ Before Being Prescribed the Drug
- Internet Access to Prescription-Free Finasteride ‘Is a Serious Risk,’ Warns PFS Investigator Roberto Melcangi‘My colleagues are seeing many cases’ of this condition, the UniMi endocrinology professor notes in his first-ever long-form TV interview Jan. 7, 2024 Dear Friends:… Read more: Internet Access to Prescription-Free Finasteride ‘Is a Serious Risk,’ Warns PFS Investigator Roberto Melcangi
Doctors & Researchers Speaking Out
Two subjects—eight percent—committed suicide during or after the study.

While the sexual side effects of 5ARIs are well known, there may be persistent genitourinary, physical, psycho-cognitive, anti-androgenic and penile vascular changes after 5ARI discontinuation. Use of 5ARIs for treatment of AGA may lead to persistent sexual, genitourinary, physical, psycho-cognitive, and anti-androgenic sequelae even after cessation of 5ARI therapy… Two subjects (8%) committed suicide during or after the study.
—Penile vascular abnormalities in young men with persistent side effects after finasteride use for the treatment of androgenic alopecia: Translational Andrology and Urology, April 2020
The new patient alert cards aim to raise awareness among men taking finasteride about the potential for psychiatric and sexual side effects.

It’s crucial that patients are aware of vital information about the medicines they’re taking. The new patient alert cards aim to raise awareness among men taking finasteride about the potential for psychiatric and sexual side effects, so they can make an informed decision about their treatment and know what to do if they experience these side effects. If you’re taking finasteride 1 mg (Propecia) for hair loss and develop depression or suicidal thoughts, stop treatment and contact your doctor.
—Safety Review of Finasteride: April 2024
[T]he pharmaceutical industry has been extremely reticent to explore this.

There is clear evidence that the symptoms of some patients who have been exposed to finasteride are unexplained, and the pharmaceutical industry has been extremely reticent to explore this. Add in relational difficulties plus premature hair loss and you could have a perfect storm to make some people more vulnerable.
—The Telegraph: May 22, 2024
[T]opical finasteride is not approved as a medicinal product in the USA.

It’s still unclear to what extent the lower systemic [finasteride] exposure translates into a lower risk of side effects. Although sexual side effects did not occur more frequently than with placebo…due to the small patient collective…hardly any reliable statements can be made. However, such side effects must always be expected. Although topical finasteride is not approved as a medicinal product in the USA, it is marketed as an over-the-counter medicinal product through telemedicine portals such as forhims.com. In this context, the Post-Finasteride Syndrome Foundation, when asked by the DAZ editors, points out that they are also aware of numerous cases of [PFS from] topical application.
—Hair Growth with Consequences: Deutsche Apotheker Zeitung, March 10, 2023
Patients & Loved Ones Crying Out
-
Mother of Patient
He was a happy, easy going, kind and caring teenager and Propecia has made him mentally disabled.
My son, 31, had a mild form of BDD and was worried about his hair thinning and took Propecia in 2013. He was taking it for 4 years during which his personality changed beyond recognition and I suspect he developed PFS. He experienced severe side effects: insomnia, loss of libido, obsessive thoughts, anxiety, depression and suicidal ideation. These deteriorated since he stopped taking Propecia in 2017. He was no longer able to go to work, lost the ability to sleep well, was unable to care for his 2 daughters and is feeling increasingly suicidal. He had to go to the emergency department twice this year and now is in a mental hospital in New York. Vulnerable young people should not be prescribed Propecia so easily and drug producers should provide a clear warning about such dangerous side effects. I believe Propecia ruined his health, career and life. He was a happy, easy going, kind and caring teenager and Propecia has made him mentally disabled. I’m very upset and do not know what to do.
GL, 62, Oxford, England: Aug. 3, 2018
Condition update: September 2020
No improvement
-
Mother of Patient
He’d been using topical medications for 5 years and decided to try Finesteride. That was the beginning of a nightmare.
My son was a healthy, smart ambitious 28-year-old man who was going back to school at Suffolk University with a 4.0. He also worked and was in fantastic health, climbing mt Washington in 4 hours for fun. He was, however, battling losing his hair. He’d been using topical medications for 5 years and decided to try Finesteride. That was the beginning of a nightmare. His sister was getting married Alugust 18th. 4 days before the wedding it started…muscle spasms, nonstop and severe anxiety. He had only been on it for 3 months. He immediately stopped taking it but it has continued for the last 5 months. Now though, he is in pain. He has been seeing a neurologist, and is convinced he must have ALS, although so far the tests do not point to that. He started seeing a therapist also hoping it was stress. But the pain and twitching and insomnia are real. He does not currently have any erectile issues thankfully but the physical and mental stress he experiences is horrible. He continues to search foir answers but i am convinced it is from the Finesteride.
LM, Lowell, MA: Jan. 6, 2019
Condition update: August 2020
Slightly worse
-
PFS Patient (male)
I am living now for seven years with my parents in their tiny apartment and do not know what to do.
I am 35 years old and have this Syndrome since I have been 24 years old. My Life was smashed and destroyed after I started with Finasteride. I can’t stop thinking about this one moment I decided to go to this special Doctor who treats hairloss. I was 24 years young and I had full beautiful hair. This Doctor made some tests and scared the shit out of me my prognosing a full bald me in a few years. I trusted him and took the pills he gave me. Safe pills he said. No side-effects. Soon after I started the pills my life made a u-turn. I was finishing university and had already found a new job in a new city. That’s when I start to enter hell. I lost my mind and had the most disgusting mental health issues. I had to go to the hospital and started with suicide thoughts. It was an absolute nightmare! Fast Forward. I am living now for seven years with my parents in their tiny apartment and do not know what to do. I feel a bit better because I take a lot of different medications but I can not go outside or even read a book because I feel sedated. I really do not know what to do with my life.
HH, 35, Stuttgart, Germany: Feb. 3, 2019
Condition update: September 2020
No improvement